|
Similar to the other electron displacement effect, mesomeric effect is also classified into positive mesomeric effect (+M or +R) and negative mesomeric effect (-M or -R) based on the nature of the functional group present adjacent to the multiple bonds. The resonance hybrid is represented as below. The resonating structures are called canonical forms and the actual structure lies between these three resonating structures, and is called a resonance hybrid. Actually, the n electrons are delocalized as shown below. This property cannot be explained by a simple structure in which two n bonds localized between C 1 – C 2 and C 3 – C 4. In 1,3 butadiene, it is expected that the bond between C 1 – C 2 and C 3 – C 4 should be shorter than that of C 2 – C 3, but the observed bond lengths are of the same. This phenomenon is also called mesomerism or mesomeric effect.įor example, the structure of aromatic compounds such as benzene and conjugated systems like 1,3 – butadiene cannot be represented by a single structure, and their, observed properties can be explained on the basis of a resonance hybrid. Such structures are called resonance structures (canonical structures) and this phenomenon is called resonance. Certain organic compounds can be represented by more than one structure and they differ only in the position of bonding and lone pair of electrons. The difference in energy between structure 1 or 2 or 3, (most stable canonical structure) and structure 4 (resonance hybrid) is called resonance energy.The resonance is a chemical phenomenon that is observed in certain organic compounds possessing double bonds at a suitable position. It is found that the energy of the resonance hybrid (structure 4) is lower than that of all possible canonical structures (Structure 1, 2 & 3). Resonance is a mental exercise and method within the Valence Bond Theory of bonding that describes the delocalization of electrons within molecules. However, the following structure gives a qualitative idea about the correct structure. Some molecules have two or more chemically equivalent Lewis electron structures, called resonance structures. 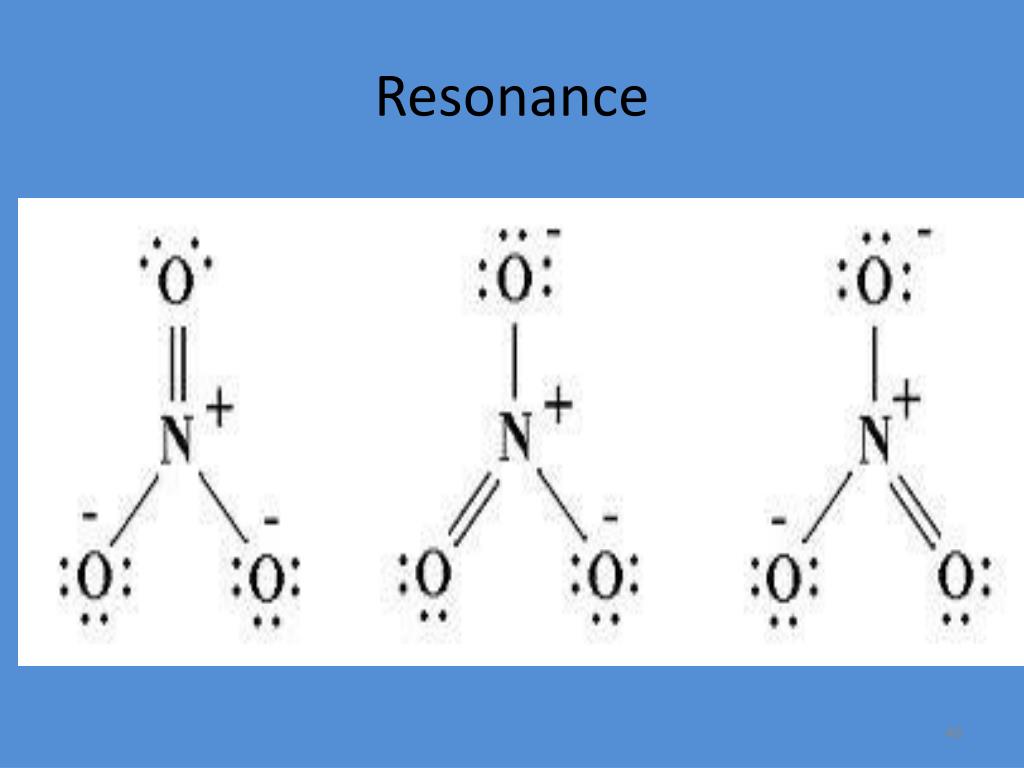
It is not possible to picturise the resonance hybrid by drawing a single Lewis structure. It is important to note that carbonate ion does not change from one structure to another and vice versa. The actual structure of the molecules is said to be the resonance hybrid, an average of these three resonance forms. It is evident from the experimental results that all carbon-oxygen bonds in carbonate ion are equivalent. Such structures are called resonance structures (canonical structures) and this phenomenon is called resonance. Resonance in chemistry is a tool used to represent and model certain types of non-classical molecular structures.Resonance is a key component of valence bond theory and arises when no single conventional model using only even number of electrons shared exclusively by two atoms can actually represent the observed molecule. They only differ in the position of bonding and lone pair of electrons. 
In this case, we can draw two additional Lewis structures by moving the lone pairs from the other two oxygens (O B and O C) thus creating three similar structures as shown below in which the relative position of the atoms are same. 
Total number of valence electrons = + + = 24 electrons.ĭistribution of these valence electrons gives us the following structure.Ĭomplete the octet for carbon by moving a lone pair from one of the oxygens (O A) and write the charge of the ion (2-) on the upper right side as shown in the figure. The skeletal structure of carbonate ion (The oxygen atoms are denoted as O A, O B & O C For example let us consider the Lewis structure of carbonate ion 2. When we write Lewis structures for a molecule, more than one valid Lewis structures are possible in certain cases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
